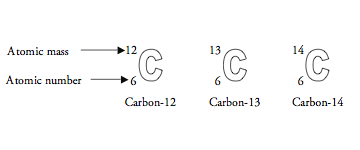 
Phosphorus is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. It is also used to construct ATP and GTP. Oxygen is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. Nitrogen is used to construct the basic building blocks of life, such as amino acids, nucleic acids, and proteins. Hydrogen is also used to construct ATP and GTP. Hydrogen is used to construct the molecules water and organic compounds with carbon. Carbon is also used to construct the energy-rich molecules adenosine triphosphate (ATP) and guanosine triphosphate (GTP). Even the cell membranes are made of proteins. Carbon is an important element for all living organisms, as it is used to construct the basic building blocks of life, such as carbohydrates, lipids, and nucleic acids. The symbol describes carbon-12 as follows: C indicates the atom is Carbon Z 6 indicates carbon-12 has 6 protons A 12 indicates that the mass number is. These elements form the basic building blocks of the major macromolecules of life, including carbohydrates, lipids, nucleic acids and proteins. The first four of these are the most important, as they are used to construct the molecules that are necessary to make up living cells. They are carbon, hydrogen, nitrogen, oxygen, phosphorus, and sulfur. In biology, the elements of life are the essential building blocks that make up living things. Carbon-12 is the common isotope, with carbon-13 as another stable isotope. 14 has 8 neutrons to remember that number of protons are same, which are 6 protons in all 3, so i hope tisu. Find step-by-step Chemistry solutions and your answer to the following textbook question: A carbon atom has a mass number of 12 and an atomic number of 6. The element is determined by the atomic number 6. Atomic number atomic number with 6 protons, 6 protons, but neutrons or the number of neutrons are different. 
Now remember that number of protons does not change and therefore in carbon 12 carbon 13 and carbon 13 have seen. Their mass number is equal to the number of protons plus the number of neutrons. The number of protons is equal to the number of protons present in an atom and if the atom is neutral, it is also equal to the number of electrons. So now, let's assume that is some atom, and here z is representing atomic number and a is representing mass number that is representing atomic number and he is representing mass number now. So here in this question, first we'll be talking about what are isotopes, so isotopes have same atomic number seem atomic number of an atom, but different mass number different mass number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
